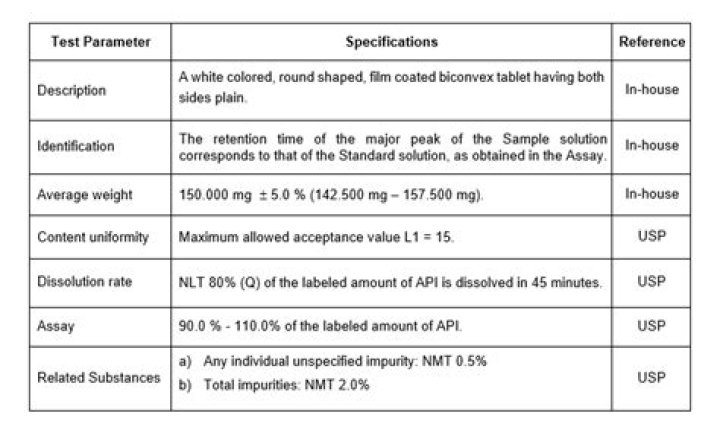
The dissolution specification is expressed in terms of the quantity (Q) of active substance dissolved in a specified time, expressed as a percentage of the content stated on the product label.
What are the two types of dissolution?
Flow Through Cell It is made of two types which are the open system and the closed system.
What is dissolution of a drug?
Dissolution is the process in which a substance forms a solution. Dissolution testing measures the extent and rate of solution formation from a dosage form, such as tablet, capsule, ointment, etc. The dissolution of a drug is important for its bioavailability and therapeutic effectiveness.
What is QC media in dissolution?
A QC dissolution method is designed to detect variations during routine product manufacture and changes during product storage that might negatively impact product performance. The key purpose of a QC method is to confirm lot-to-lot consistency of commercial product at release and over the shelf life of the product.
How do you determine dissolution?
Dissolution (with rate dm/dt) occurs from a solid with mass = m and surface area = A, from the saturation concentration at the particle surface (Cs) to the concentration in the bulk solution (Cb).
Why do we use sinkers in dissolution?
Floating dosage forms rarely dissolve at a reproducible rate. Using a sinker in Apparatus 2 fixes the dosage form to a certain point and aids reproducibility. Sinkers can also be useful where dosage forms may stick to the sides of the vessel.
How do you perform a dissolution test?
Place the stated volume of the dissolution medium, free from dissolved air, into the vessel of the apparatus. Assemble the apparatus and warm the dissolution medium to 36.5° to 37.5°. Unless otherwise stated, place one dosage unit in the apparatus, taking care to exclude air bubbles from the surface of the dosage unit.
How many types of dissolution are there?
There are seven USP-defined types of dissolution apparatus: baskets, paddles, reciprocating cylinders, flow through cells, paddle over disk types, cylinders, and reciprocating holders. Although USP 2 paddles are most widely used, most dissolution apparatus incorporate any number of each type (often all of them) .
What is the difference between DT and dissolution?
Disintegration is a process of breaking down a substance into tiny fragments to improve its solubility in a solvent. Dissolution, on the other hand, is a process through which solutes dissolve in a solvent. Dissolution is also used predominantly in pharmaceutical industries to check how soluble a drug is in the body.
Why do we use paddle in dissolution?
It is cylindrical in shape and is constructed from non-reactive mesh to prevent any unwanted chemical reactions that may alter the final result. The pores in the mesh allow the dissolving drug to move from the basket into the holding vessel, creating a uniform solution.
What is the difference between solution and dissolution?
As nouns the difference between solution and dissolution is that solution is a homogeneous mixture, which may be liquid, gas or solid, formed by dissolving one or more substances while dissolution is the termination of an organized body or legislative assembly, especially a formal dismissal.
What is Q value in dissolution?
USP defines Q as the quantity or the “amount of dissolved Active Pharmaceutical Ingredient (API) specified in an individual monograph, expressed as a percentage of the labeled content of the dosage unit…”. When we look at a Q value, we are looking at what percent has dissolved at that time for that product.
Why dissolution test is required?
Dissolution testing is a requirement for all solid oral dosage forms and is used throughout the development life-cycle for product release and stability testing. It is a pivotal analytical test used for detecting physical changes in an active pharmaceutical ingredient and formulated product.
What are the types of dissolution?
Defined dissolution apparatus and development of new tools Currently, there are seven different types of dissolution apparatus defined in the United States Pharmacopeia (USP)-basket type, paddle type, reciprocating cylinder, flow through cell, paddle over disc, rotating cylinder, and reciprocating disc.
What does Q mean in dissolution?
The quantity, Q is the amount of dissolved active. Dissolution Medium—Proceed as directed for Immediate- ingredient. specified in the individual monograph, expressed Release Dosage Forms under Apparatus 1 and Apparatus 2.
How does a dissolution work?
Dissolution is the process where a solute in gaseous, liquid, or solid phase dissolves in a solvent to form a solution. Solubility is the maximum concentration of a solute that can dissolve in a solvent at a given temperature. At the maximum concentration of solute, the solution is said to be saturated.
How does pH affect dissolution rate?
Solubility is Affected by pH By changing the pH of the solution, you can change the charge state of the solute. If the pH of the solution is such that a particular molecule carries no net electric charge, the solute often has minimal solubility and precipitates out of the solution.
What is S1 S2 S3 criteria for dissolution?
In stage S3, another 12 units are to be analyzed, the average of 24 units (S1+S2+S3) should be equal to or more than Q and no unit is less than Q-25% and not more than 2 units are less than Q-15%.
Why dissolution is performed on 6 tablets?
Learn about the dissolution stages followed during the failure of the sample using the six dosage form units. Dissolution test is done to verify the release of drug in the solution from the tablet because of binders, granulation, mixing and the coating may affect the release of drug from tablets.
Capsule Sinkers. Floating dosage forms rarely dissolve at a reproducible rate. Using a sinker in Apparatus 2 fixes the dosage form to a certain point and aids reproducibility. Sinkers can also be useful where dosage forms may stick to the sides of the vessel.
Solvent is generally a liquid that is used to dissolve the solute. Solution is referred to as the mixture that results from dissolving the solute in solvent. ‘Solubility’ is a characteristic property of the solute and ‘Dissolution’ is the process in which a solute dissolves in a solvent to result a solution.
Is there a general chapter for dissolution testing?
General Chapter <711> Dissolution has the specifics for dissolution testing methodology. There are articles that provide more dissolution development background. Skoug et al. (2) gave one of the first comprehensive overviews of the development, validation, and specification setting for dissolution testing.
Which is an example of a dissolution method?
It is expressed as percentage release of drug substances present dosage forms such Tablets, Capsules, oral suspensions and ointments. In our present study dissolution method development for various kinds of tablets such as immediate release or instant release, modified, Extended release and Enteric coated. [1] [2] [3]
What should be the dissolution level of a drug?
Some start at low dissolution levels (e.g., 25%–40% at 15 minutes), whereas others start at approximately 75% at 15 minutes. The result is a collection of dissolution profiles with a variety of response patterns. Experiments are commonly conducted to study the effects of various factors on the dissolution profile.
What are the properties of a dissolution profile?
In Mandel’s model, each “row” of the data table is adissolution profile resulting from a set of experimental conditions. dissolution profiles have two general properties: level and shape. The ideal approach would be to have a single statistic to measure level and a second statistic to quantify shape.